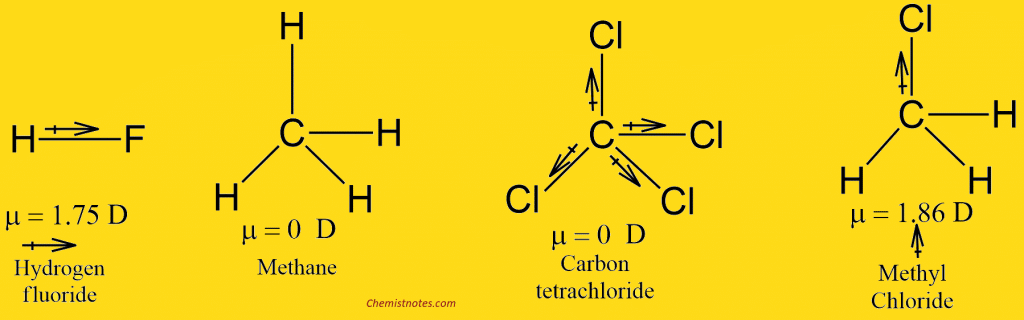
Molecules with an inversion center, such as ethene (C 2H 4) and sulfur hexafluoride (SF 6), do not have a permanent dipole moment because all three components of the dipole change sign under inversion (are non-invariant). This is true not only for molecules in an A 1 state, but holds for molecules in any non-degenerate state (a state that transforms according to a one-dimensional irreducible representation of the point group). Only the components of the dipole that transform according to A 1 are non-vanishing. Most molecular ground states transform as A 1 (alternatively expressed as: "the symmetry species of the ground state is A 1"). This point group has irreducible representations among which is the totally symmetric one, commonly denoted by A 1. More technically: the symmetry operations of a molecule that is rigid-i.e., the nuclei are clamped in space, but the electrons "move" in the quantum mechanical sense of the word-form a group, the point group of the molecule. This is a contradiction and, hence, either a dipole component is zero or it is invariant, i.e., does not change under any of the symmetry operations. If a dipole component would change under a symmetry operation, it would give a means to distinguish the old from the new conformation, so that the two conformations would be distinguishable. By definition a symmetry operation changes a molecule to a conformation that is indistinguishable from the original conformation. This rule can be proved formally, but can also be understood intuitively. For the same reason the dipoles in the examples lie along the (twofold) rotation axis of their molecule. As an example, the rule explains why the molecular dipole moments in figure 1 (water) and in figure 2 (orthodichlorobenzene) are in the vertical and horizontal mirror plane of their molecule reflection in a plane that contains a dipole leaves the dipole invariant. Provided a molecule is in a totally symmetric state, it can be shown that a dipole component vanishes when it is not totally symmetric (not invariant under all symmetry operations). Usually one considers molecules in their ground (lowest energy) electronic state and this state is almost always totally symmetric, i.e., invariant under all symmetry operations. Spectroscopic measurements and quantum chemical calculations have confirmed the linearity of CO 2. 
Since it has not, its structure is in all likelihood linear. For instance, if the gas phase molecule CO 2 were bent, like H 2O, it would have an observable dipole. Conversely, if measurements show that a molecular dipole is zero, it gives an indication of the symmetry of the molecule. Symmetry arguments can explain the vanishing of components of a molecular dipole (symmetry gives a " selection rule"), but symmetry arguments cannot directly predict the magnitudes of the non-vanishing components. Whether a molecule has one or more non-zero permanent dipole components depends on the symmetry of the molecule and on the symmetry species of the molecular state under consideration. Dipole moments can be computed reliably for smaller molecules (say up to 10 second-period atoms) by means of quantum chemical methods. Molecular dipole moments may be obtained experimentally the main techniques are microwave spectroscopy and measurements of dielectric constants as function of temperature. The figure shows the dipole moment of water in the physics convention. For physicists the direction of the dipole vector is from negative to positive charge and for many chemists it is the opposite, namely from positive to negative charge. However, all scales predict the same direction of the dipole. These concepts are semi-quantitative and different scales for electronegativity and electropositivity of the chemical elements are in use, leading to differing values of molecular dipole moments. An electronegative atom attracts electrons (becomes negative) and an electropositive atom donates electrons (becomes positive). In chemistry, polarity is usually explained by the presence of electronegative and/or electropositive atoms in the molecule. The size of this dipole, referred to as the molecular dipole moment, gives an indication of the polarity of the molecule, that is, the dipole moment is a measure for the amount of charge separation in a molecule. Because of symmetry the vector has one non-vanishing component it points from the negative to the positive region of the molecule.īeing a charge distribution, a molecule may possess a permanent electric dipole, a vector known as the molecular dipole. The molecule is electrically neutral: 2δ + + δ 2 − = 0.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
